
Quality Manual ISO 9001:2015 Quality Management System
A Quality Manual is a document that outlines the quality management system (QMS) of an organization. It provides an overview of the organization's quality policies, objectives, and processes, following the requirements of the ISO 9001:2015 standard. ISO 9001:2015 is an internationally recognized standard that sets the criteria for implementing a quality management system in an organization, focusing on enhancing customer satisfaction and achieving continuous improvement.
Introduction:
In today's competitive business landscape, maintaining consistent quality in products and services is crucial for a company's success. To achieve this, organizations often adhere to internationally recognized quality management standards such as ISO 9001:2015. This article delves into the significance of a Quality Manual in the context of ISO 9001:2015 and highlights its role in establishing a robust Quality Management System (QMS).
Understanding ISO 9001:2015:
ISO 9001:2015 is a globally accepted standard developed by the International Organization for Standardization (ISO). It sets out the criteria for implementing a Quality Management System that can help organizations enhance customer satisfaction, improve internal processes, and drive continual improvement. This standard focuses on various aspects, including risk-based thinking, leadership involvement, and a process approach to quality management.
200+ Premium Ecommerce Courses Free ❤️
Free Courses on Amazon FBA, Facebook Marketplace, and More
The Importance of a Quality Manual:
A Quality Manual serves as a central document within the ISO 9001:2015 framework. It acts as a guide that outlines an organization's QMS structure, policies, procedures, and processes. The manual provides a comprehensive overview of how the company plans, executes, and controls its operations while ensuring adherence to ISO standards.
Components of a Quality Manual:
- Introduction and Scope
This section provides an introduction to the Quality Manual, including the purpose, scope, and applicability of the QMS within the organization. - Quality Policy
The Quality Policy is a statement of the organization's commitment to quality and customer satisfaction. It outlines the organization's overall quality objectives and sets the framework for quality-related activities. - Organizational Context
This section describes the internal and external factors that influence the organization's QMS, including its stakeholders, industry context, and the needs and expectations of interested parties. - QMS Structure
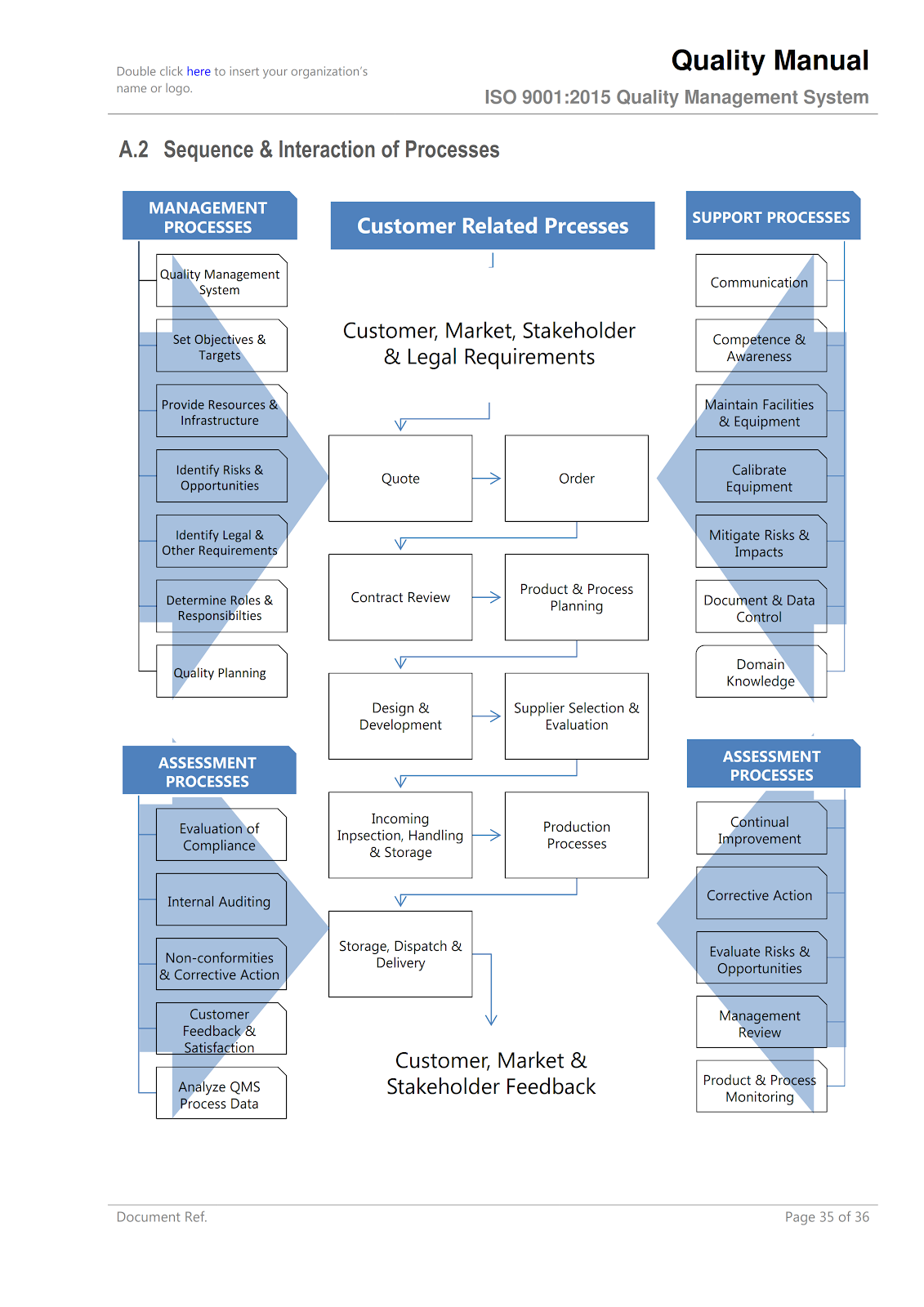
Here, the organization presents an overview of its QMS structure, including the processes and their interactions. This might include flowcharts or diagrams to illustrate the QMS. - QMS Processes
Each process within the QMS is detailed separately, including their purpose, inputs, outputs, responsibilities, and interactions. These processes cover activities like management review, resource management, product realization, measurement and monitoring, and improvement. - Roles and Responsibilities
The Quality Manual defines the roles and responsibilities of key personnel involved in the QMS, including top management, process owners, and other relevant positions. - Document Control
This section outlines the procedures for document control, including the process for document creation, review, approval, distribution, and revision. - Record Keeping
The Quality Manual describes the requirements for the retention and maintenance of records related to the QMS processes. - Monitoring and Measurement
The organization outlines the methods and criteria for monitoring and measuring the QMS processes and performance. - Non-Conformance and Corrective Action
This section explains the process for identifying and addressing non-conformances and taking corrective actions to prevent recurrence. - Continual Improvement
The Quality Manual highlights the organization's commitment to continual improvement of the QMS, including processes for identifying opportunities for improvement and implementing corresponding actions. - Management Review
This section outlines the process for conducting management reviews of the QMS, where top management evaluates its effectiveness and makes decisions on improvements.
Quality Manual ISO 9001:2015 Quality Management System
Quality Manual ISO 9001:2015 Quality Management System
Implementing the Quality Management System:
To successfully implement the QMS, organizations need to define and document the processes while ensuring their compliance with ISO 9001:2015 requirements. Employee training and communication play a vital role in fostering a culture of quality and ensuring that everyone understands their roles in maintaining high standards.
Training and Communication:
Employees at all levels should receive appropriate training to understand the QMS, their responsibilities, and how they contribute to achieving quality objectives. Effective communication channels facilitate the dissemination of information and feedback throughout the organization.
Performance Evaluation:
To measure the effectiveness of the QMS, regular performance evaluations are essential. Internal audits help identify areas for improvement and compliance gaps, while management reviews assess the overall performance of the QMS.
Benefits of ISO 9001:2015:
Implementing ISO 9001:2015 offers numerous advantages to organizations, including:
- Enhanced customer satisfaction through improved product and service quality.
- Increased operational efficiency and productivity.
- Reduced waste and costs through streamlined processes.
- Better risk management and mitigation strategies.
- Increased credibility and trust among stakeholders and customers.
- Improved decision-making based on data-driven insights.
Conclusion:
A Quality Manual is a cornerstone of ISO 9001:2015 implementation, guiding organizations towards effective quality management. By creating a well-structured manual and diligently adhering to the ISO standards, businesses can achieve a competitive edge, build customer loyalty, and drive continual improvement. Embracing ISO 9001:2015 demonstrates a commitment to excellence and positions companies as leaders in their respective industries.
FAQs:
- What is ISO 9001:2015?
ISO 9001:2015 is an internationally recognized standard that outlines the requirements for a Quality Management System. - Why is a Quality Manual important?
A Quality Manual provides a comprehensive overview of an organization's QMS, ensuring consistent processes and adherence to ISO standards. - How can ISO 9001:2015 benefit my business?
ISO 9001:2015 can enhance customer satisfaction, increase efficiency, reduce costs, and improve overall performance. - What is risk-based thinking in ISO 9001:2015?
Risk-based thinking involves identifying and addressing potential risks to prevent negative impacts on the QMS. - Is ISO 9001:2015 applicable to all industries?
Yes, ISO 9001:2015 is applicable to organizations of all sizes and industries seeking to improve their quality management practices.






0 Comments